Fresh from vacation, ex-McKinsey analyst and then-venture capitalist Corey McCann M.D. Ph.D. stepped up to the mic at the first-ever NeuroGaming conference in 2013 and asked a question. Well, three
9.13.19
17 min. Read
The Pear Therapeutics Report
In this article:
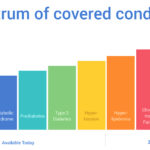
In this 4,500-word report, E&O digs into the strategic moves, challenges, and successes that the Pear Therapeutics executive team has navigated since its founding five years ago. The report covers dozens of Pear’s therapeutic areas of interest; financial details from licensing agreements the company has forged with academic researchers; why it acquired a small tech startup in its early years; its failed direct-to-consumer products and more.
×
![]()