Issue 074
Get E&O weekly. | Subscribe
Digital health research from Brian Dolan.
Welcome to E&O.
Last week I wrote about Germany’s first two reimbursed prescription digital therapeutics and how pharma execs think digital products should be priced. Here’s what’s happening this week:
- (OK, this was earlier in the month but) Fitbit quietly snagged a $900,000 contract with the Department of Veterans Affairs. The project is called: “Early Detection and Long-term Impacts of COVID-19 Identifying Risks and Resilience with Digital Health Technologies.” Remember, the VA works closely with Google, which is still in the legally-complicated process of digesting Fitbit.
- Meanwhile, the FDA cleared LIVMOR’s continuous AFib monitor as an Rx-only device. Why it’s interesting: It runs on a Samsung smartwatch. “The LIVMOR AF detection system is FDA cleared class-II medical device software. It is powered by the LIVMOR Halo + home monitoring system, an FDA registered medical solution that is provided to the patients pre-loaded and validated on a Samsung Gear S2 with an integrated PPG monitor. Samsung Gear S2 are not FDA cleared for the detection of Atrial Fibrillation in the absence of LIVMOR controls.”
- Update: Zocdoc responded to its founding CEO’s public comments surrounding his lawsuit against the company, which I mentioned in September. In a blog post, the company added more color to its past and present financial performance: “In 2015, Zocdoc generated $71 million in annual revenue. However, we also burned through $43 million in cash that year. Despite this high burn rate, revenue had flat-lined, growing by just one percent month-over-month by October 2015.”
- Another Headspace Health lead departs: Pablo Pantaleoni, the VP and GM of Headspace’s digital therapeutics subsidiary Headspace Health, has left the company to join behavioral health provider LifeStance Health as Chief Digital Officer. (Considering the company’s false start into DTx this kind of thing makes me wonder how it’s going over there?)
- And another mySugr CEO departs Roche: Anton Kittelberger, a mySugr co-founder and longtime COO turned CEO, left to start a new thing with his mySugr co-founder, Frank Westermann. Roche acquired mySugr in 2017.
- If you haven’t gotten enough explanation of what went wrong (and right!) at Proteus Digital Health, the company’s two founders are out with dueling LinkedIn posts this week. Here’s Andrew Thompson’s. Here’s Dr. George Savage’s. They mostly repeat (or slightly build on) what I wrote last week.
- Medical journal Neurorehabilitation and Neural Repair published results from an early MedRhythms feasibility study, which carried a pithy title: “A Music-Based Digital Therapeutic: Proof-of-Concept Automation of a Progressive and Individualized Rhythm-Based Walking Training Program After Stroke.”
- Worthwhile study in Nature Digital Medicine from voice biomarker company Aural Analytics: “Early detection and tracking of bulbar changes in ALS via frequent and remote speech analysis.”
- One more thing… Boehringer Ingelheim announced a digital health-focused study for heart failure patients that it is conducting with Yale. The study will use a smart scale from Bodyport, live-coaching from Noom, and automated nudges from Conversa. Read the announcement here (https://www.boehringer-ingelheim.us/press-release/boehringer-ingelheim-and-yale-clinical-and-translational-research-accelerator) or the trial’s page over at clinicaltrials.gov here.
Thanks for being an E&O subscriber. If this was forwarded to you from a friend, let’s assume they think you should be a paying subscriber too. Don’t think too much about it: Click here to get the full E&O experience.

FDA’s 2021 plan puts risk categories for SaMD and new guidance for opioid use disorder devices on the “maybe” list
As it does each year, the agency just published its list of planned final and draft guidances for FY2021, which it breaks down into A and B lists. The A list is the real plan, while the B list includes ones they hope to get to if they have the resources.
If you remember anything from the year 2019, you might recall I wondered last November why the FDA didn’t have anything about artifical intelligence in its 2020 plans (Issue 026).
Two of the potential guidances the FDA might get to on its B list include:
- A draft guidance on: “Risk Categorization for Software as a Medical Device: FDA Interpretation, Policy and Considerations.”
- A draft guidance on: “Clinical Considerations for Medical Device Premarket Submissions Targeting Opioid Use Disorder.”
That second one is particularly curious considering two of the few prescription digital therapeutics on the market today are focused on OUD.
The A list has a number of interesting items, including:
- A final guidance on clinical decision support software.
- A draft guidance for a “Transition Plan for Medical Devices Distributed Under Enforcement Policies or Emergency Use Authorization (EUA) During the COVID-19 Public Health Emergency.”
Read the full list here and send the agency feedback.
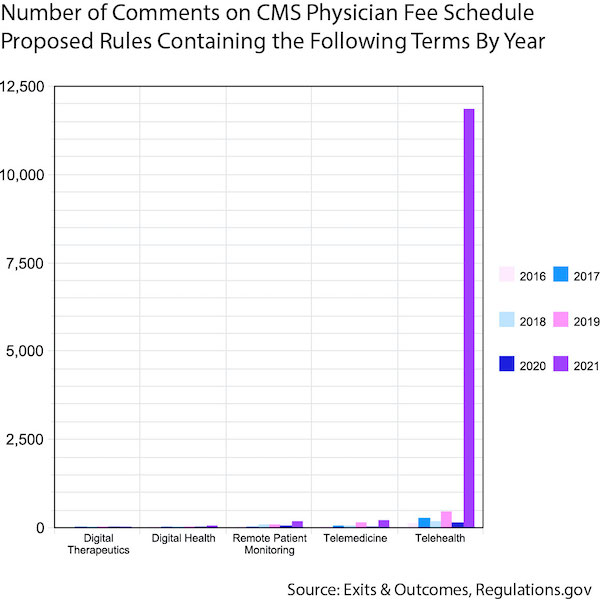
Industry comments on the annual CMS Physician Fee Schedule proposed rule show “telehealth” spike
The comment period for The Centers for Medicare & Medicaid Services (CMS) proposal for the FY2021 Physician Fee Schedule closed during the first week of October.
It’s no secret that telehealth has boomed in recent months because of the COVID–19 pandemic.
The graph above shows how quickly telehealth became a part of the conversation when it comes to CMS reimbursement. Incredibly, more than 11,870 comments sent to CMS in response to its FY2021 reimbursement plans contained the word “telehealth”. That’s up from just 156 comments using the word last year in response to the FY2020 Physician Fee Schedule and the previous record-holder — 455 comments contained the word “telehealth” in response to the FY2019 proposed rule.
“Digital therapeutic(s)” got a mention in a whopping eight responses to the FY2021 proposed rule this year, which points to this submarket’s still budding reimbursement effort.
Updates to the Prescription Digital Therapeutic Pipeline of Pipelines
This week I added six products from Amalgam to the Prescription Digital Therapeutic Pipeline of Pipelines Database, which you can read through here. Who am I missing?
One name that readers have sent my way a few times is the pioneering digital therapeutic Freespira from Palo Alto Life Sciences.
Freespira consists of a handheld tablet that comes pre-loaded with software that shows patients specific breathing protocols. It also includes a device that measures their breathing rate and exhaled CO2 in real-time. A Freespira coach or the patient’s therapist then uses that data to remotely coach and train them.
Freespira is not technically a prescription therapeutic, however. As the DTx Alliance’s page on the product explains:
“Authorization from a licensed healthcare provider is required. A prescription from a physician is not necessary… Patients may self-refer into the program or are referred by a healthcare provider. The Freespira Patient Care Team determines any necessary copay/coinsurance payments, collects payments or creates a flexible payment plan, then ships devices to the patient’s home and schedules training.”
Quick links: E&O research reports and databases
The links below aim to make it easier for paying subscribers to find the long-form research reports and databases on the E&O site:
The Virgin Pulse Report (Subscribers-only Link)
The Evidation Health Report (Subscribers-only Link)
Database: Rx-only Digital Therapeutics Pipeline of Pipelines (Subscribers-only Link)
Database: Online Diabetes Prevention Program Companies (Subscribers-only Link)
Database: Digital Health PPP Loans (Open Access)
The Proteus Digital Health Report (Subscribers-only Link)
The Hinge Health Report (Subscribers-only Link)
The Digital Health Enrollment Report (Subscribers-only Link)
The Omada Health Report (Subscribers-only Link)
The Google Health Report (Subscribers-only Link)
The Pear Therapeutics Report (Subscribers-only Link)
The AliveCor Report (Subscribers-only Link)
Apple’s Healthcare Work Experience (Subscribers-only Link)
Approximating Livongo’s S-1 (Subscribers-only Link)
That’s a wrap on Issue 074 of E&O.