Issue 003. FDA Pre-Cert Dropouts? Amazon wearable.
Get E&O weekly. | Subscribe | Digital health research from Brian Dolan.
FDA wants more Pre-Cert participants
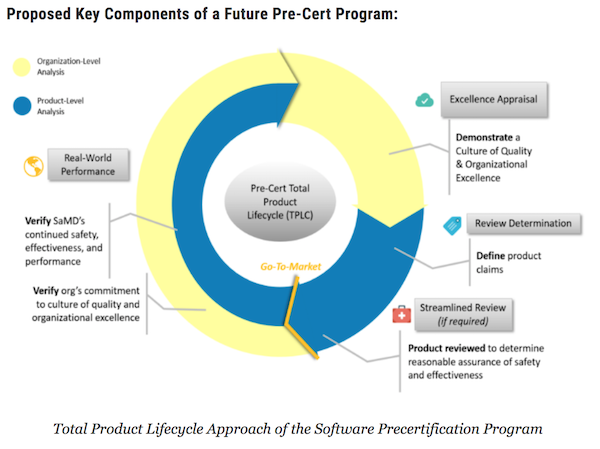
In something of a surprise move, the FDA announced this week it would open its Pre-Cert program up to new participants who will be accepted on a rolling basis for what it's calling the 2019 Test Plan. The first batch of Pre-Cert participants had to show they were planning to apply for a De Novo clearance, but this new call for volunteers seeks both De Novo and 510(K) applicants. One industry insider (and longtime Pre-Cert detractor) wondered whether the news means some of the original nine participants dropped out. As a reminder, those included: Apple, J&J, Fitbit, Pear Therapeutics, Phosphorous, Roche, Samsung, Tidepool, and Verily.
Rock Health follow-up analysis to its Q1 funding report
"Since Q1 2018, the top five deals each