PLUS: Six digital pharma studies
Issue 081
 E&O Fridays.
E&O Fridays.
Welcome back. Last week in E&O Fridays, I wrote about PDTs vs digital vaccines (?) vs disease management 2.0 and dug into the Mahana Therapeutics De Novo’s evidence base.
On Wednesday, I had a bit of a longer Fortune 500 Digital Health Stack piece, because I tried to tackle JPMorgan Chase, which sent me down a Haven rabbit hole. E&O Wednesdays also mentioned Omada’s 2020 marketing strategy and Noom’s increasing interest in self-insured employers.
Finally, on Monday, I wrote about last week’s more than $600 million in announced and unannounced health tech funding, raised an eyebrow at the digital health mention in Salesforce’s explanation for its pricey acquisition of Slack, and served up a brand new (much more difficult) Health Tech Trivia Challenge. I’ll reveal the three winners Monday.
But first, here’s what’s happening this week in the world of digital pharma products and FDA-regulated digital health:
- Last week in my write-up of Mahana Therapeutics‘ De Novo clearance, I made an assumption that the company delivered its IBS prescription digital therapeutic in the form of a smartphone app. Since then the company made its official announcement, and Parallel is actually a web app and intended for use on a laptop or desktop computer. That’s important. It likely means the academic-developed predecessor, a web app named Regul8, is close to identical to Mahana’s web app, Parallel. So, Mahana may not face the same challenges Pear Therapeutics did when it came under fire from ICER for equating a web app used in a clinic to a mobile app used anywhere.
- Speaking of Pear, it just raised an $80 million Series D led by Softbank. Wow.
- Google launched a quasi-competitor to Apple ResearchKit called Google Health Studies that works on Android phones. The company launched it with an initial focus on respiratory illnesses like COVID and flu. Seems to be mostly survey-based, patient-reported symptoms in this first iteration.
- The German government is about to add yet another digital health offering to its fast track reimbursement directory, DiGA. According to Newsenselab, its migraine-focused digital therapeutic M-sense Migraine will be added to the directory. (It’s not in there as of this writing though.)
- Meanwhile, Roche inked a deal with Diabeloop: The partnership “marks Roche’s first step in the field of automated insulin delivery (AID) representing a milestone in Roche’s strategy of integrated Personalised Diabetes Management (iPDM).”
- More than a month ago I reported that Palo Alto Life Sciences, which is now doing business simply as Freespira, raised a new round. This week the news is official: Freespira, maker of an FDA-cleared digital therapeutic for panic attacks, panic disorder PTSD raised $10 million led by Lightspeed Venture Partners.
- Japan-based CureApp said it just became the first prescription digital therapeutic company to get reimbursement from that country’s insurance system. CureApp’s first product is focused on nicotine cessation. If prescribed by a doctor, patients will only have to pay less than 30 percent of the price of the therapeutic.
- Weeks after its FDA authorization, PTSD and nightmare-focused prescription DTx Nightware inked a deal with First Nation Group, which is a longtime medical supplies contractor with the federal government.
- Former Cognoa CEO turned venture capitalist Brent Vaughn has taken the helm as CEO of Alzheimer’s-focused, clinical-stage DTx company, Cognito.
- Paris-based Tilak Healthcare, a digital therapeutics company that offers a mobile game, OdySight, to monitor and rehab aging-related chronic eye diseases, announced an expanded agreement with Novartis Pharma to co-promote the DTx in markets outside of France too.
- One more thing… Longtime tech writer Benedict Evans got me thinking about global pharma companies going digital and digital health companies going global with these two tweets: “An increasingly important concept in tech regulation — ‘the highest common denominator’. This is when a jurisdiction passes a law whose legal or engineering implications mean that if you want to operate in that jurisdiction, you have to apply that law globally. See GDPR, CCPA. Hence, the toughest applicable law that’s been passed anywhere your global footprint has to be followed *everywhere* in your global footprint — it’s the highest common denominator.”
If this was forwarded to you from a friend: Click here to sign yourself up as a paying subscriber. Then, you too can win friends and influence people via helpful newsletter forwards.

Akili Interactive to price EndeavorRx at $450 for three months of video game-based ADHD treatment
On a recent podcast produced by MM&M, Akili Interactive’s Chief Marketing Officer Meghan Rivera, who just joined the company in September, shared a bit more about Akili’s plans as it brings EndeavorRx to market.
Here are a few excerpts from the interview:
“We are not reliant on payer coverage to grow volume. We have priced ourselves competitively. The out-of-pocket cost for caregivers, even before we have coverage, is incredibly reasonable. And we are seeing incredibly low abandonment due to cost, which, out of the gates, is a really positive thing to see, because we know other digital therapeutics companies have struggled in this arena. We didn’t want it to be a gating factor or a rate limiter for us from a commercial perspective. ”
Pricing: $450 for three months, less than $100/month for cash pay
“We have come to market with a $450 list price. There is an opportunity right now, because we don’t have payer coverage to any broad extent, to mark that down for cashpay patient to less than $100 per month course of therapy. So, that $450 is for three months as a list price.”
Akili also has a patient assistance program (PAP) for lower-income patients who qualify. Rivera noted that there is a significant ADHD patient population in the US on Medicaid.
About the launch: “It won’t be traditional”
“I can tell you this [about the commercial launch of EndeavorRx], it won’t be traditional. We are basically re-imagining the entirety of the commercial model, which is why this opportunity, for me personally, has been so exciting. It is a blank slate. We have the opportunity to launch a digital therapeutic into the market and prove out that we can be incredibly successful in the short term.”
Rivera said that Akili’s team is “all-in” on experimenting and iterating with its product, but it plans to take a similar approach to its commercial strategy.
“How do we think about things differently? We don’t necessarily need a sales team of several hundred people knocking on healthcare provider doors. Has anybody tested that model? Why wouldn’t we be exploring how to manage and build a sales function in a different kind of way? Especially during COVID times, but I would argue even outside of COVID when things maybe come back to at least a new normal following a vaccine. But, yes, I think it will be something that the world hasn’t necessarily seen before even from other prescription digital therapeutics companies.”
Rivera said that part of the strategy will likely include “a heavy focus on consumer activation” because Akili believes this to be a caregiver-driven market. She also said that, given the safety profile of EndeavorRx, there is a very low-risk to prescribing this therapeutic, Akili will focus on a digital channel to market.
Reading between the lines a little: It sure sounds like Akili will set up a telemedicine-based prescription channel like Pear Therapeutics recently announced it would do for its insomnia DTx, Somryst.
Rivera said that a telemedicine approach would work not only because the product is digital and needs to be downloaded anyway, but also because there are so many care providers as part of a child’s ADHD care team. Not all of them have the ability to write prescriptions, depending on which state they practice in.
So, if a cognitive behavioral therapist working in a state where they don’t have the authority to write prescriptions wants one of their patients to try EndeavorRx out, Akili wants to provide them with a “Bat Phone” that allows them to connect to someone who can write the Rx.
Listen to the whole 30-minute-plus podcast over at MM&M’s site here.
FDA clears Apple ECG 2.0 and AliveCor sues Apple for alleged patent infringement
Apparently, back in October, the FDA cleared a new version of Apple Watch’s ECG app, which Apple is calling ECG 2.0 App in its regulatory filings.
The new ECG 2.0 App uses the original’s De Novo clearance as its predicate. It has a few added capabilities, including a number of new results it can display related to high heart rate readings:
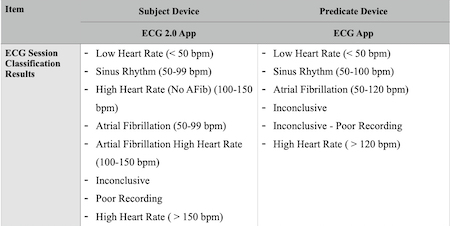
Similarly, its indications for use is almost identical to the 1.0 ECG App, save for the bit about high heart rate and its accompanying parenthetical aside below:
“The ECG app determines the presence of atrial fibrillation (AFib), sinus rhythm, and high heart rate (no detected AF with heart rate 100-150 bpm) on a classifiable waveform. The ECG app is not recommended for users with other known arrhythmias.”
Weirdly, no one noticed this 510(k) clearance in the FDA database until yesterday. (I asked the FDA to confirm precisely when they added it because I’m having a hard time accepting that I missed it in there. I check that database almost every day! I’ll let you know when I hear back.)
The delayed timing of the world noticing this development also coincides with a related patent infringement lawsuit.
At the beginning of the week, AliveCor filed a patent infringement lawsuit in Texas that alleges Apple’s ECG App violates three of AliveCor’s patents:
“Apple’s infringement has damaged and continues to damage AliveCor in an amount yet to be determined, but at least a reasonable royalty and/or the lost profits that AliveCor would have made but for Apple’s acts of infringement.”
Now I’m curious if Fitbit and Samsung — and other wearable makers who added ECG detection to their wares in the years since Apple did — will also hear from AliveCor’s lawyers?
Digital Health studies: Novartis, Pfizer, AstraZeneca, Proteus, Bold Health, and Galderma
One of my favorite Thursday night activities is combing through the Clinical Trials DOT gov database in search of studies I think E&O readers should know about. Here’s a quick run-down of what I found this week:
Novartis Breezehaler study with Propeller Health: This is a big study. They’re aiming for 1,000 participants across Germany: “This study is designed as a multicenter, observational, non-interventional, open label, 26-week study in order to observe how asthma control changes under treatment with Mometasone Fuorat/Indacaterol/ Glycopyrronium (MF/IND/GLY) Breezhaler® sensor system or under treatment with fixed-dose combination (FDC) triple therapy after 26 weeks of treatment.” Study
Scripps’ 10-person Proteus Digital Health study: This one just finished up in August but the results were only posted in mid-November. The study included 10 heart patients with implanted assistive devices who took a total of 40 doses of Proteus sensor-enabled medications. Some 80 percent of those doses were detected by the Proteus system within the allotted 30 minute time period. Study
Bold Health’s Zemedy vs Headspace for IBS: Bold Health is set to launch an experimental study that will pit its digital therapeutics for IBS, Zemedy, up against Headspace: “The research method is experimental, cross over (randomized controlled trial) with a control control. Participation in the study will last approximately 4.5 months, depending on the subjects willingness to participate and rate of completion.” Basically, depending on the arm a participant is assigned to, they may end up using Zemedy or Headspace or both. Is that unusual to use a competitor’s intervention in a study like this? Seems risky — what if Headspace shows better results? Study
Galderma’s moisture-sensing device and app rosacea study: This study just got pushed back from an October 2020 start date until January 2021, but it will make use of a handheld, Bluetooth-enabled device that uses sensors to track moisture in their skin and then sends that data to an app on their smartphone. These devices are available for $250 online. Galderma, a pharmaceutical company focused on skin diseases, and its research partners will equip about 30 participants with these GPSkin Barrier devices: “The primary hypothesis is that weekly digital interactions and routine measurement of TEWL rates and SC hydration levels will promote patient adherence to maintenance ivermectin therapy and prevent disease relapse.” Study
AstraZeneca using wearables to try to detect pulmonary acute exacerbations: The pharmaco is using wearables, tablets, mobile apps, connected spirometers in this 75-person lung cancer patient study: “Data collected will be used to evaluate the likelihood of early detection of pulmonary AEs in unresectable Stage III NSCLC patients on durvalumab. This project seeks to understand if multiparametric mobile technology collecting patient-reported outcomes, vital signs, and respiratory function, integrate well into a patient’s daily life and aid physicians in early detection of pulmonary AEs.” Study
Pfizer studies the LyfeMD app for Ulcerative Colitis and Crohn’s: The app offers users customized anti-inflammatory diet plans, exercise plans, as well as mindfulness and stress reduction programs. The name of the 44-person RCT is: “Using the LyfeMD Application for Effective Maintenance of Remission and Improved Quality of Life in Crohn’s Disease and Ulcerative Colitis: A Randomized Controlled Trial.” Study
Wow, that escalated quickly. I’ll try to write less next week. So long, Issue 081 of E&O Fridays.